I had a nice visit from code enforcement the other day. They said that my "work bench" is not allow in plain view. Evidently "work" is the operative word here, when it was a "bar" it was perfectly allowable. So I have to keep the tools out of sight or camouflage them with spiced rum labels. Interesting illustration of perspective.
Astronomers look at a planet and determine by the radiant energy that that planet has a specific temperature. Eout has to be equal to Ein, so they know every thing they need to know about that planet. Engineers look at a system and know that Ein is about equal to Eout but there is work going on inside so they peal back the spiced rum label to see what is going on inside. Energy is fungible, but the work done is not, if the mechanism to perform the work is not there, the work doesn't get done. So some scientist are more like code enforcement than engineers, they see what they think they see, not what is really there.
Inside where all the good stuff happens tells the tale. The ocean is a huge mass of water capable of hole huge quantities of energy. That energy is contained in all directions or the energy would quickly dissipate. So how is the energy stored, converted and manipulated in the system until it gets to the spice rum label?
Water has a specific heat capacity. It freezes at a temperature around 0 degrees and boils at a temperature around 100 degrees. It requires 4.2Joules of energy to raise the temperature of water one degree. So to change the gram of water from 0 to 100 degrees requires 420Joules of energy plus of minus a touch.
To freeze water, you have to remove 334Joules per gram. To melt water, you would have to add the 334Joules per gram. That happens at one temperature for both processes if the water is pure, at two different temperatures if the water is not pure. Each degree of change in the freezing point to the melting point requires another 4.2Joules per gram of energy. To evaporate water requires 2257Joules per gram at the boiling point of water which also varies a little depending on the purity.
If the whole range of energy is used from freezing to boiling, a total of approximately 2257+420+334=3011Joules per gram is required. once the water freezes or evaporates, it is no longer in the ocean, it either floats on the surface or is transferred to the atmosphere. Either way it in no longer in the physical ocean of liquid. If there is no atmosphere the water cannot evaporate and if the water were tightly contained, the container would either expand or burst. So the volume of water would have to expand or contract if the energy did not remain constant. For the water cycle, the oceans and atmosphere are fully coupled, meaning a change in one will cause a change in the other.
If I have used the right values this time, the range of temperature for pure water is from 273.15K (0C) to 373.15K (100C) degrees. Note that 273.16K (0.01C) is more exact, but close enough that I should not have to redo all my drawings :)
The Stefan-Bolztmann Black Body Equation, E=5.67x10^-8(T)^4 is the relationship of the energy emitted by an object at temperature T in degrees Kelvin. By using the freezing and boiling points of water, the energy range is 1099.3Wm-2 for 100C and 315.6Wm-2 for 0C, a range of 783.6Wm-2 per 100C or 7.84 per degree C. You may note that this is difference than 4.2Joules per gram where Joules per sec is equal to Watts per second by definition in Systems International units. Why would that be?
If we had one gram of water spread thinly over a 1 meter square surface between a body of water and space it would emit 7.84Wm-2 outwardly but 4.2Wm-2 inwardly. It is easier for the energy to flow into space than it is to warm water. Warmer water has greater potential energy, so the energy of that thin layer of water would do less work and (7.84/4.2=1.86) 86% more escaping. If the energy flow into the water was equal to the energy flow to space the work done would equal the energy lost, the process would be 50% efficient. Considering the addition of an atmosphere instead of space, up to 86% more work would be done on the atmosphere than on the water. The energy is only 14% efficient warming water.
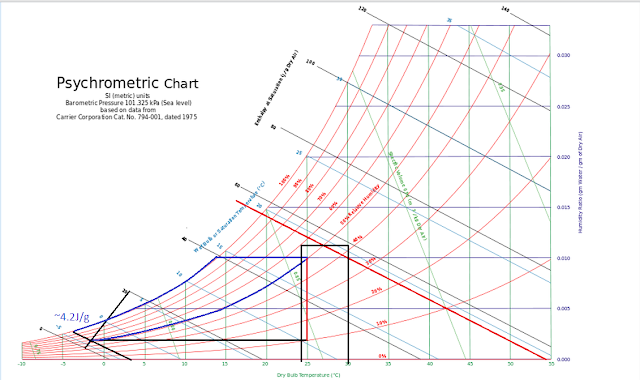
The atmosphere has a specific heat capacity. 1.006Joules per gram of dry air at 25C and 1.013 bars, sea level. This is close to 1 so to simplify, water has 4.2 times the heat capacity of dry air at sea level and 25 degrees C. If all of the energy of our thin gram of water was transfer to both the air and water. The air would gain 86 percent more of the 4.2 Joules, .86*4.2=3.16 Joules per gram and .14*4.2=0.59 Joules per gram for water. Under this steady state condition, it would not take long for all the energy in the water to be lost to the atmosphere. For equilibrium, the water has to absorb the same energy as the air, water is virtually incompressable and air is not, so the air would have to absorb more energy and expand or more water vapor to increase its specific heat capacity or both. At sea level, the point were moist air reaches 4.2Joules per gram is approximately -1.9C and 50% relative humidity. That point varies from approximately -3C for saturated air and to +3C for dry air.

With moisture available because of the ocean and expansion available, limited by gravity, the psychrometric chart provides a reasonable range of conditions inside of the moist air boundary. The work performed on the atmosphere would be a combination expansion and thermal capacity change. The work performed on the ocean would be mainly change in the thermal capacity with some limited expansion. The work performed on each would be equal at equilibrium.
Since we live on a water world, water has been around for perhaps a billion years, there is a pretty good chance that there is some conservation of water involved in the thermodynamics of the system. There is a pretty good deal of ice on Earth. We know that ice releases 334joules per gram as it forms so it is somewhat likely that the energy gained would balance energy released. Using the Wm-2/Joules per second relationship, the maximum energy allowed in equilibrium would be 316Wm-2 plus 334Wm-2 released from the formation of fresh water ice or 650Wm-2 which has an equivalent temperature 327.2degrees K (54.1 degrees C) +/-3 degrees for dry air. For the water, the energy lost to evaporation would have to be considered.
Since the psych chart is posted above, a simple approximation is just to follow the 50% RH curve until it intersects with the enthalpy of dry air at 54.1C degrees, then add the -3 to +3 range of the lower enthalpy limit.
As shown, that results in a maximum SST range of 24C to 30C, which is a rough estimate, but generally close enough for government work. To refine that estimate we can look at the latent heat of evaporation, 2257J/g remembering that we are assuming conservation of the water on the planet.
As ice forms it releases 334J/g of energy. If that energy is released inside of the moisture boundary, some portion would be transferred to the atmosphere and some to the ocean to regain equilibrium. When water evaporates it absorbs 2257J/g of energy which is then contained in the atmosphere. If ice is being formed and evaporation taking place without any reverse of these processes, there would be no liquid water on Earth. There has to be an equilibrium if liquid water is to be conserved. When ice is forming and water is condensing, both release energy to the system. Since the ocean can only absorb 14% of the energy released, 86% would be lost from the system. If ice melts and water vapor condenses, the system would absorb all that energy, the system would run away. Over some period of time, the enthalpy released would have to equal the enthalpy gained to conserve both energy and water. Since the energy of the heat of evaporation is much larger than fusion, it would be the limiting process which can be dealt with by the assumption of a constant relative humidity. Using the Psych chart we can extent the 50%RH curve to the maximum enthalpy at 54.1C. Follow that line to saturation, the down the saturation curve to where there is half the initial enthalpy between 4.2J/g, the minimum and approximately 56J/g the maximum, then to the right on the sensible heating line back to 50%RH, that results in an approximate average surface temperature of 21.5C degrees. Probably close, but enough uncertainty not impress many.

Another way is to consider the energy of the moisture boundary temperature which at 0C would be 316Wm-2. That energy is provide by the sea surface and only 86% of the sea surface skin layer is transmitted to the atmosphere in equilibrium. So the effective radiant energy would be 316/.86=367.4Wm-2 or 10.6 degrees C. That temperature is the saturation temperature on the chart above which when sensibly heated to 50%RH again results in an estimated surface temperature of 21.5C degrees.
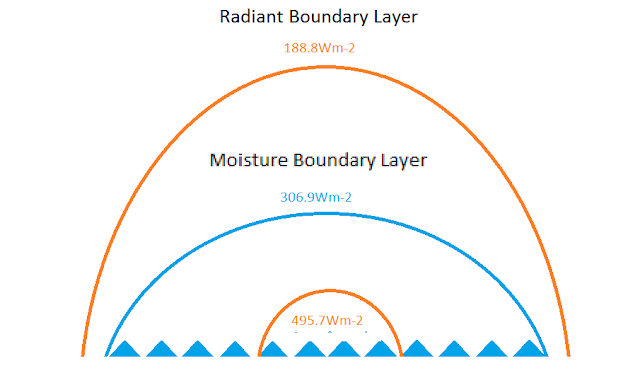
We could even abandon the psych chart and just consider the energy required to balance the heat of fusion of the ice, 334J/g. Since the ratio of the heats fusion and evaporation is 334/2257=0.1497, only 15% of the mass of ice formed is the needed mass of water evaporated to maintain equilibrium. So the average surface temperature would be 86% of the distance between the minimum temperature and the maximum temperature. The maximum temperature is only estimated, so there is a way to estimate the change solely on the 334 J/g, .86*344=287.24Wm-2 plus the initial energy 316Wm-2 would equal 603Wm-2 if the energy was perfectly transferred and contained. The atmosphere outside of the moisture boundary envelope is far from a perfect container, so energy would be lost in the transfer. The 603Wm-2 would decrease to equilibrium with the surface and the moisture boundary envelope at 316Wm-2. What limit it would decay or reduce to would depend on the energy balancing the 316Wm-2 at the moisture boundary.
This part may be tricky for some. For the moisture boundary layer to be in equilibrium with space, there must be an equal and opposite balancing force. 50% efficiency is the maximum for this steady state system as mentioned earlier with the 4.2 into the ocean surface versus the 7.84 to space. At the moisture boundary layer with 316Wm-2 energy, it would be balanced by half with half lost to space. The 603 would decay by approximately 158Wm-2 to 445Wm-2 if the moisture boundary layer were 316Wm-2. We have a range of values for the moisture boundary layer though, +/-3C at the boundary. The point previously plotted on the psych chart at 50% relative was approximate -1.9C which has an equivalent energy of 307Wm-2 which would be balanced by 153.5Wm-2. For fresh ice to form in a salt ocean it has to release the 334J/g for fusion plus 1.9*4.2=7.98 addition J/g to reduce the temperature by 1.9 C degrees. The 603 would have to decay an additional 7.98 plus 4.5 or 12.5Wm-2 to 445-12.5=432.5Wm-2 which would have an equivalent temperature of 22.5 C degrees. The decay would not be perfectly linear nor is the boundary efficiency estimate perfect, but again this should be close enough for government work.
So what does all this BS mean? Basically, that as long as there is liquid water on Earth with sufficient thermal mass, the ocean average temperature will be in the range of 22.5 C +/- a couple of degrees. The total area of the open water ocean would vary, the volume of the moist air envelope would vary, but the average temperature of the ocean is surprisingly stable due to the thermal properties of water and moist air. A point completely glossed over in the global warming debate.
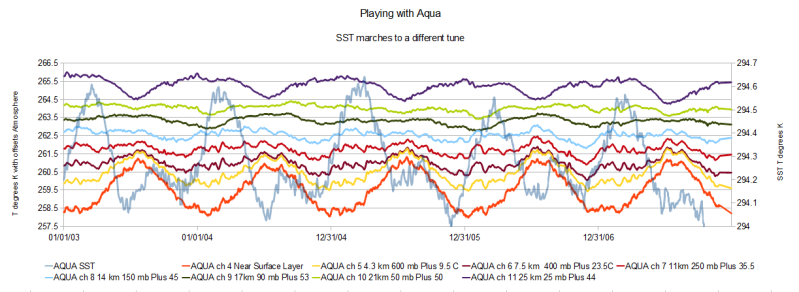
Think of what this simple model projects, a maximum surface temperature of 54.1C, Aziziya, Libya has a maximum record temperature of 57.8C that has never been repeated with the next maximum list as 54C in Israel. The average SST range via the AQUA satellite is approximately 294.25K degrees 21.1C degrees, 1.4C less than the model estimate that made a linear assumption for an energy decay. These are actual values compared to a crude model that does not seriously consider any radiant impacts or even mass, volumes or input energy. Any significant radiant impact from Greenhouse gases will be felt outside of the moisture boundary envelope which will considerably reduce any current estimate based on radiant only impact looking in from the spiced rum sticker :)
I haven't completely proofed this, but since it was raining today, felt like posting a different perspective.